Sexually transmitted infections
Gonorrhoea is among the three most common sexually transmitted infections, and its resistance to antibiotics is rapidly spreading. There is now only one remaining last-resort treatment for gonorrhoea. If we continue down the current path, this treatable disease may become untreatable. GARDP is acting now to develop a new antibiotic for gonorrhoea infection in patients with limited treatment options.

Programme goals

A new, accessible treatment for gonorrhoea, a disease that affects 82 million people globally each year.

Explore different interventions for STIs, in line with GARDP’s 2024-2028 Strategy.
“To keep pace with the inevitable emergence of drug resistance to the few treatments currently available, we must invest now in developing new antibiotics, researching further existing antibiotic regimens, and accelerating equitable antibiotic access.”
– Alison Luckey, Associate Director Medical Sciences

Current project
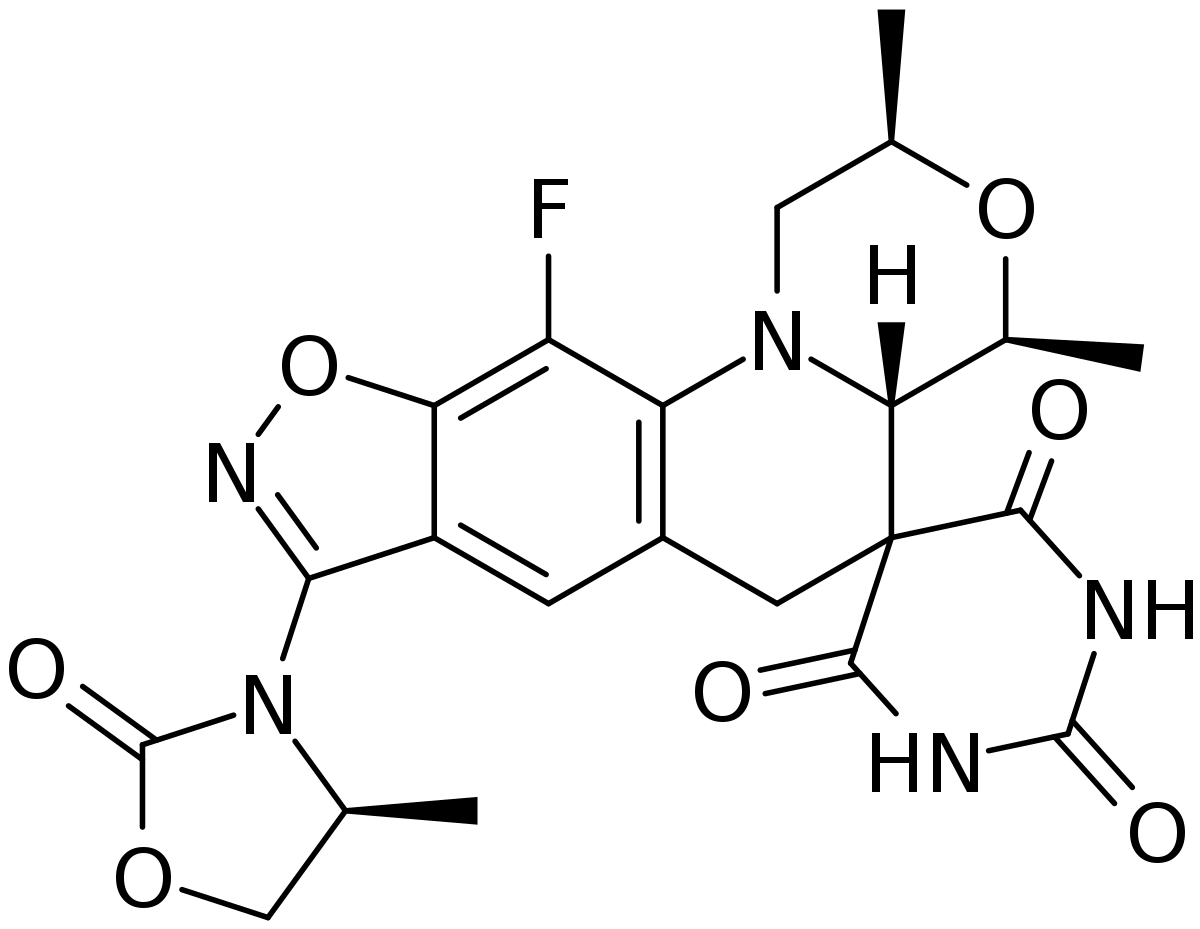
Zoliflodacin drug development project
In 2017, GARDP partnered with Entasis Therapeutics Limited, now an affiliate of Innoviva Specialty Therapeutics, to develop an antibiotic, zoliflodacin, to treat uncomplicated gonorrhoea infection in patients. The global phase 3 pivotal registration trial was carried out between 2019 and 2023 at 16 sites across five countries (Belgium, Netherlands, South Africa, Thailand and the US).
A total of 930 patients with uncomplicated gonorrhoea were recruited and randomised in the phase 3 study, making it the largest clinical trial ever conducted for a new treatment against gonorrhoea infection. It is the first antibiotic trial led and sponsored by a non-profit like GARDP.
In November 2023, the positive results of the pivotal phase 3 trial were announced. A single dose of oral zoliflodacin was found to meet the primary endpoint when compared against the combination of injectable ceftriaxone and oral azithromycin, a current international standard of care.
As a first-in-class drug, zoliflodacin has a new, unique mechanism of action. It is conveniently administered orally—rather than injected like the current standard treatment. An oral treatment like zoliflodacin also has the potential advantage of being used to treat sexual partners who do not always seek clinical care.
Under the collaboration agreement, GARDP has the rights to register and commercialize zoliflodacin upon approval in more than three-quarters of countries worldwide, including all low-income countries, most middle-income countries and several high-income countries.


Warning: Undefined array key "felt" in /home/clients/c9b0ed6b0786a56afdaf5611fa7b11b2/web/20221216/wp-content/themes/gardp2/templates/acf/parallax_mobile.php on line 11
Warning: Trying to access array offset on value of type null in /home/clients/c9b0ed6b0786a56afdaf5611fa7b11b2/web/20221216/wp-content/themes/gardp2/templates/acf/parallax_mobile.php on line 11

Warning: Undefined array key "felt" in /home/clients/c9b0ed6b0786a56afdaf5611fa7b11b2/web/20221216/wp-content/themes/gardp2/templates/acf/parallax_mobile.php on line 11
Warning: Trying to access array offset on value of type null in /home/clients/c9b0ed6b0786a56afdaf5611fa7b11b2/web/20221216/wp-content/themes/gardp2/templates/acf/parallax_mobile.php on line 11


Key milestones
- SIGNED collaboration agreement with Entasis to develop and register zoliflodacin, with the goal to provide access to patients who suffer from drug-resistant gonorrhoea in low- and middle-income countries
- ENROLLING patients at all sites in phase 3 trial of zoliflodacin
- COMPLETED manufacturing of the three batches of zoliflodacin for new drug registration
- SIGNED Memorandum of understanding with Dr Reddy’s Laboratories and Aurigene Pharmaceutical Services Limited (APSL) in India, as well as with the Thailand Ministry of Public Health, to work toward improving access to zoliflodacin upon approval
- COMPLETED prevalence study of sexually transmitted infections, including gonorrhoea, in Kenya (results forthcoming)
- COMPLETED recruitment for global phase 3 trial of zoliflodacin
- COMPLETED pivotal phase 3 trial
- ANNOUNCED topline data for phase 3 trial
- ANNOUNCED US FDA acceptance of New Drug Application for zoliflodacin





About gonorrhoea
About gonorrhoea
Gonorrhoea is a widespread sexually transmitted infection that is treatable in most cases with antibiotics. However, resistant strains of Neisseria gonorrhoeae, the bacteria responsible for gonorrhoea, are on the rise all around the world, including in early 2022 three new cases of “super-gonorrhoea” in the UK alone. The World Health Organization has thus labelled Neisseria gonorrhoeae a priority pathogen in urgent need of new treatments.
If left untreated or when untreatable, gonorrhoea can cause serious, lifelong consequences in men and women, and can amplify the spread of HIV in high-prevalence settings.
The disease disproportionately harms women, who often fail to show early symptoms and thus do not seek treatment. When left untreated in women, gonorrhoea can lead to pelvic inflammatory disease that elevates the risk of complications in pregnancy, including the likelihood of ectopic pregnancies and infertility. During birth, gonorrhoea can be transmitted to babies, who in turn may have health problems like gonococcal conjunctivitis and skin infections.